02xl is your source for accurate and trustworthy information so you can make the best choices for your health and wellness.
PS: Please note that 02xl provides this information for the benefit of the rare disease and overweight community. 02xl is not a medical provider or health care facility and thus can neither diagnose any disease or disorder nor endorse or recommend any specific medical treatments. Patients must rely on the personal and individualized medical advice of their qualified health care professionals before seeking any information related to their particular diagnosis, cure or treatment of a condition or disorder.
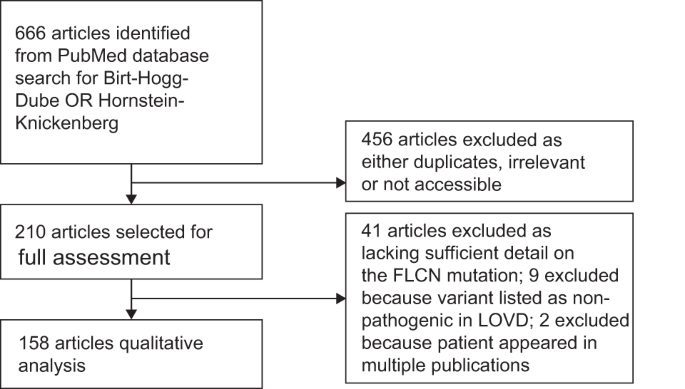
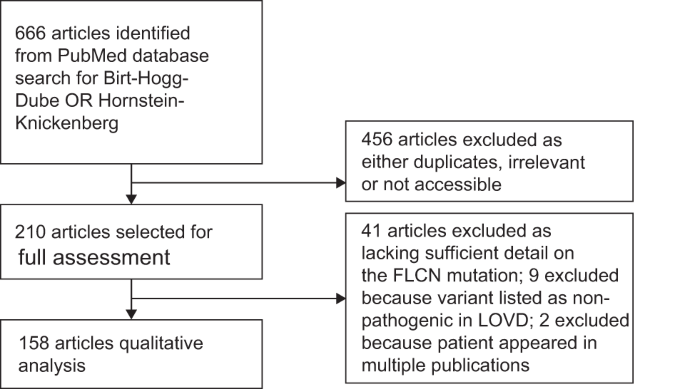
We identified 666 papers on BHDS. Of these, 158 met the inclusion criteria by reporting genetically confirmed pathogenic FLCN variants (Fig. 1). A total of 1059 individuals from 575 families were thus identified. Patients ranged in age from 2 to 92 years (median 46 years); 45.7% were male, and 68.8% had a confirmed family history of BHDS. Geographically, 32.7% were diagnosed in North America, 31.7% in Europe and 33.3% in E-Asia (Japan, South Korea, China and Taiwan).
Clinical features
Overall, 50.9% (528/1038) of reported individuals had experienced at least one pneumothorax (Table 1) [2, 4,5,6, 8,9,10,11,12,13,14,15,16,17,18,19,20,21,22,23,24,25,26,27,28,29,30,31,32,33,34,35,36,37,38,39,40,41]. Of these, 66.7% (220/330) had recurrent pneumothoraces. There was no lateralisation of the pneumothoraces (46.4% left, 53.6% right). The median age at first pneumothorax was 34 years (range 10–78 years; n = 257), while the median age of individuals at the time of their case report was 46 years (range 14–92 years; n = 342). Of the patients who underwent thoracic CT imaging, 92.1% (662/720) had reported lung cysts. Of these patients, 63.7% had a pneumothorax [2, 4,5,6, 8,9,10,11,12,13,14,15,16,17,18, 21,22,23,24,25,26,27,28, 30, 31, 34,35,36,37,38,39,40,41].
Of the 989 patients with recorded dermatological examinations, 47.9% had lesions consistent with BHDS [2, 4,5,6, 8,9,10,11,12,13,14,15,16,17,18, 20,21,22,23,24,25,26,27,28,29,30,31,32,33,34,35,36,37,38,39,40,41]. Most had fibrofolliculomas (90.5%; 429/474), 8.8% (42/474) had trichodiscomas and 4.0% (19/474) had perifollicular adenomas. The median age at which skin changes were first noted was 38 years (range 20–65 years; n = 44), while the median age of these individuals at the time of their case report was 51 years (range 22–92 years; n = 213).
Of the 929 patients who underwent imaging of the abdomen, 22.5% had malignant renal tumours [2, 6, 8,9,10,11,12,13,14,15,16,17,18, 20,21,22,23,24,25,26,27,28,29, 31,32,33,34,35,36,37,38,39,40,41]. In 52.5% (62/118), the tumour was unifocal and did not recur after excision. A variety of histological subtypes were reported: chromophobe 32.8% (63/192), hybrid oncocytoma-chromophobe 24.5% (47/192), clear cell 11.5% (22/192), oncocytomas 9.9% (19/192), hybrid clear cell-chromophobe 7.3% (14/192), papillary 5.2% (10/192) and the remaining patients had either a hybrid oncocytoma-clear cell or a hybrid papillary-clear cell tumour. The median age at first diagnosis of renal cell carcinoma was 47 years (range 14–83 years; n = 108). Their median age at the time of case report was 52 years (14–92 years; n = 121).
Differences were apparent in the phenotypes reported in geographical regions. The frequency of pulmonary cysts in East Asia was significantly higher (96.8%; 302/312) than North America (89.0%; 218/245) or Europe (88.2%; 134/152) (p < 0.0001) [2, 4,5,6, 8,9,10,11,12,13,14,15,16,17,18, 20,21,22,23,24,25,26,27,28,29,30, 34,35,36,37,38,39,40,41]. Similarly, a higher proportion of pneumothorax was seen in East Asian reports (73.9%; 249/337) compared with North America (35.1%, 120/342) or Europe (44.6%, 150/336) (p < 0.0001). By contrast, a slightly higher proportion of renal cancer was observed in European reports (27.1%, 83/306) than in either North American (23.9%, 75/314) or East Asian individuals (16.4%, 47/286) (p < 0.05). More strikingly, the proportion of dermatological features was far lower in East Asian (20.6%, 66/320) than in European (50.3%, 153/304) or North American cases (73.4%, 251/342) (p < 0.0001).
Of the individuals where the data were available, the commonest presentation was a positive family history of the disorder (44.0% of individuals, 438/995) [2, 4,5,6, 8,9,10,11,12,13,14,15,16,17,18, 20,21,22,23,24,25,26,27,28,29, 31,32,33,34,35,36,37,38,39,40,41]. Skin findings were the presenting feature in 17.8% (177/995) of diagnoses, followed by pneumothorax in 19.6% (195/995) and renal cancer in 7.5% (75/995) of cases. The remainder could not be linked to a single feature or were the result of incidental detection of pulmonary cysts after CT scanning. Of note, a longer latency to diagnosis was observed when pneumothorax was the first clinical feature (median time to diagnosis 6 years; n = 138) compared with 0 years for both RCC (n = 13) and skin changes (n = 36).
Genetic features
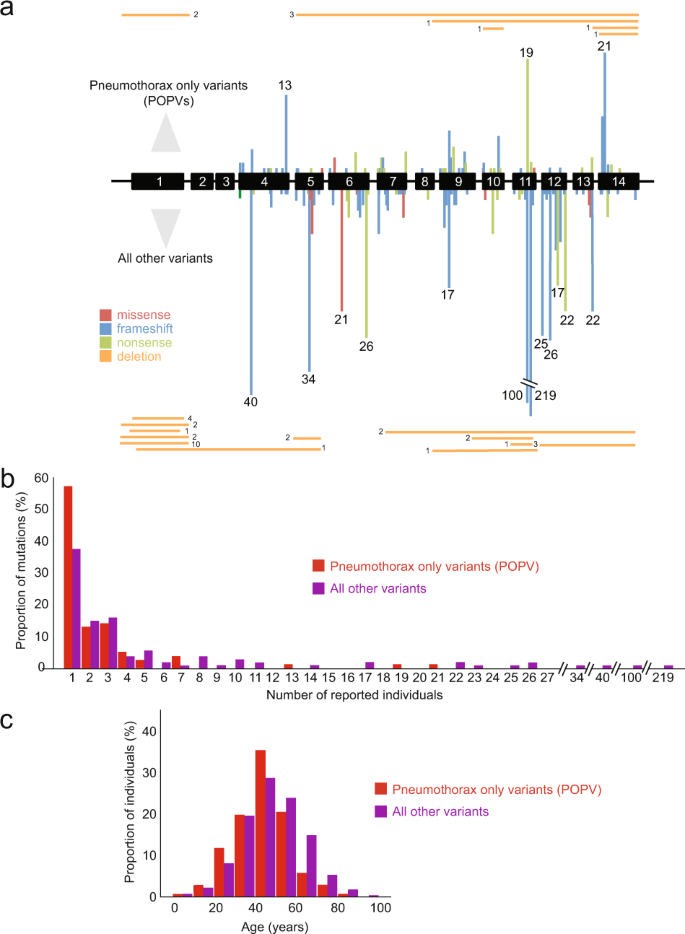
A total of 194 pathogenic variants were identified from the literature [2, 4,5,6, 8,9,10,11,12,13,14,15,16,17,18, 20,21,22,23,24,25,26,27,28,29,30,31,32,33,34,35,36,37,38,39,40,41,42]. These comprised 132 nonsense and frameshift variants, 31 intronic variants, 11 missense variants/in-frame deletions, 13 large deletions/duplications and 7 variants affecting the initiation of transcription (Fig. 2A and Supplementary Table 1). Eleven variants had no associated patient details and so were excluded from subsequent analyses.
A Missense/in-frame variants in red, frameshifts in blue, nonsense variants in light green, large deletions/duplications in orange. Bars are proportional to numbers of affected individuals, for mutational hotspots the number of individuals is given above each bar. ‘Pneumothorax-only’ pathogenic variants (POPV) shown above exons, all other variants shown below. B Histogram of individuals affected by POPV (red), all other variants (purple). C Ages of individuals reported to carry POPV (red) or all other FLCN variants (purple) (colour figure online).
It has been suggested that certain FLCN variants lead to a forme fruste of BHDS with pneumothoraces but no renal cancers [4, 5]. Since the existence of ‘pneumothorax-only’ FLCN variants would have important consequences for screening protocols, we examined these in more detail. Of the 183 variants, there were 76 ‘pneumothorax-only’ variants, 24 also had skin changes (Fig. 2A).
POPVs were distributed throughout the gene (Fig. 2A) and no association was found with variant type (p = 0.78). We hypothesised that at least some FLCN variants might erroneously appear as POPVs if detected in younger or smaller families with less chance of having manifested renal carcinomas and were not adequately followed-up (given that the median age at first pneumothorax was 34 years and median age at RCC was 47 years). Indeed, we observed that a majority of putative POPVs (85.5%, 65/76) affected no more that three individuals (Fig. 2B, C). Furthermore, the median age of individuals with POPVs was significantly lower than those with non-pneumothorax-only variants (45 vs. 47 years, p < 0.01). Importantly, there were less data on elderly members (70 years or over) in families with reportedly ‘pneumothorax-only’ variants (Fig. 2C).
Of 991 individuals studied in this review, 10 were diagnosed before the age of 18 (range: 2 to 17) and a further 7 were diagnosed between the ages of 18 and 20 [8, 12, 16, 18, 33,34,35,36,37,38,39,40]. Of the ten patients diagnosed before the age of 18, six were asymptomatic (five diagnosed through family screening and one through a mutation analysis following a diagnosis of leiomyosarcoma), three presented with pneumothoraces and one presented with renal cell carcinoma. Overall, there were 14 patients with a FLCN variant, who had their first pneumothorax before the age of 20 [8, 12, 13, 18, 31, 32, 34,35,36, 41]. There was one case of renal cell carcinoma [33] and no cases of pathognomonic skin lesions. The youngest ages of presentation for pneumothorax and renal cell carcinoma were 10 and 14, respectively [33, 36].